The agency has accepted over 1,600 applications, with the majority being for e-cigarette or e-liquid products.Read More
But the re-categorization does not appear to extend to nicotine vapor products.Read More
The distributor will assist in the rollout of VLN cigarettes in Colorado. Read More
AVM says its members alone have received more acceptance letters than the FDA admits. Read More
The agency has also sent more than 340 warning letters to manufacturers and retailers. Read More
The debut follows strong performance of the brand during its pilot in Chicago. Read More
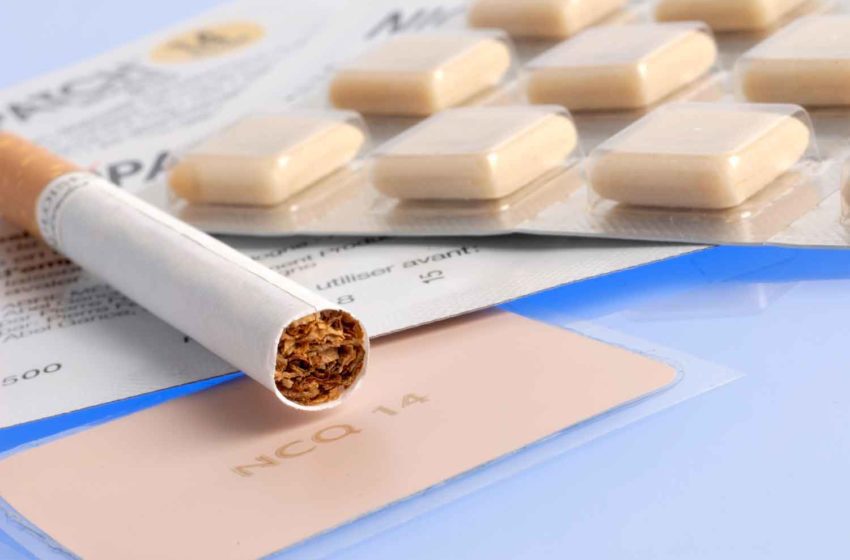
The availability of nicotine with minimal harm justifies a rethink of our approach to this drug.Read More
The FDA is processing applications for some 1 million non-tobacco nicotine products.Read More
The project explores how best to minimize the nicotine strength in vaping products.Read More
The agency will face greater pressure to prove it's authorized to pursue its agenda. Read More
Recent Posts
- Brazil Volume Down, Earnings Up
- Vaping Tied to Reduced Smoking Prevalence
- FDA Almost Done Reviewing Mass Market Products: Report
- Russia: Share of Smokers Halved
- Azerbaijan Hikes Tobacco Taxes
- BAT Reports Results
- Kutsaga Releases Climate Resilient Seeds
- Tracking Legislation Advances in Philippines
- BAT Pays Fine in Nigeria
- Regulator Proposes Retail Licensing
- KT&G Volunteers Help With Harvest
- IQOS’ U.S. Launch Postponed
- Zimbabwean Cigarette Exports up by Half
- Smoke-free Product Sales Boost PMI Income
- Air Global Launches R&D Facility in Dubai