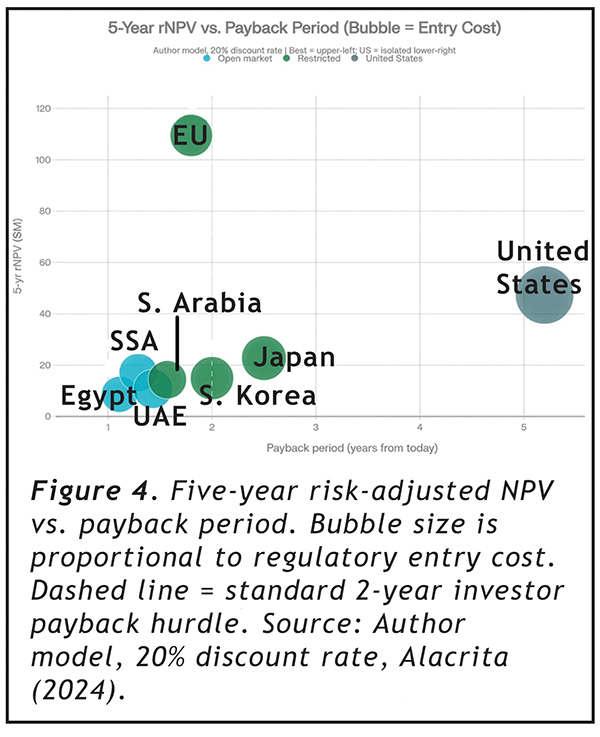
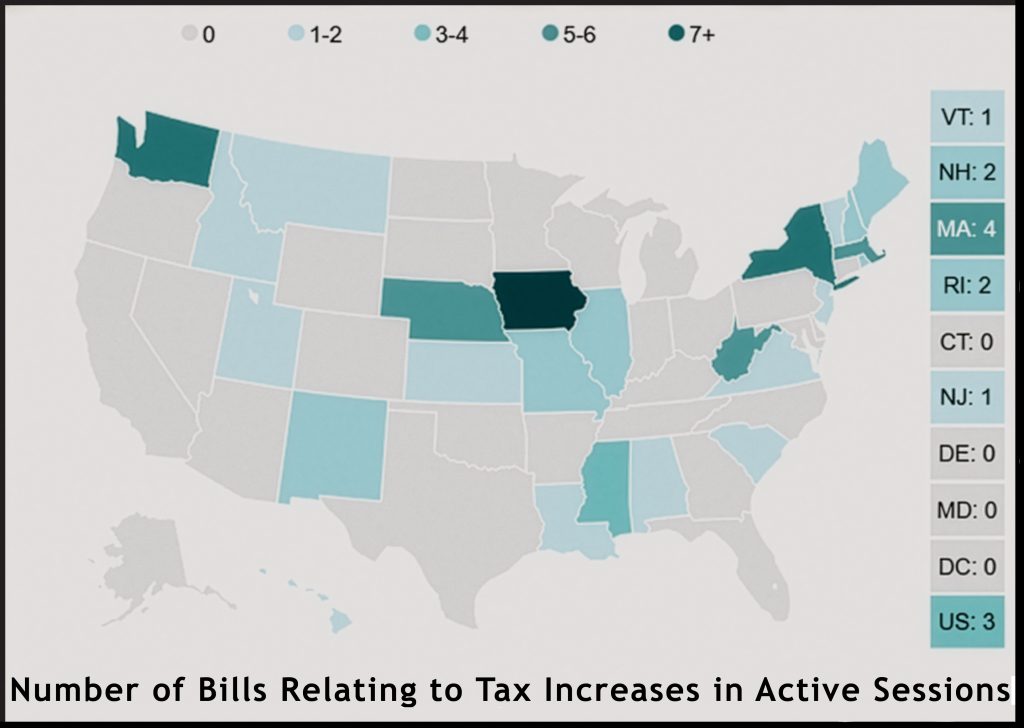
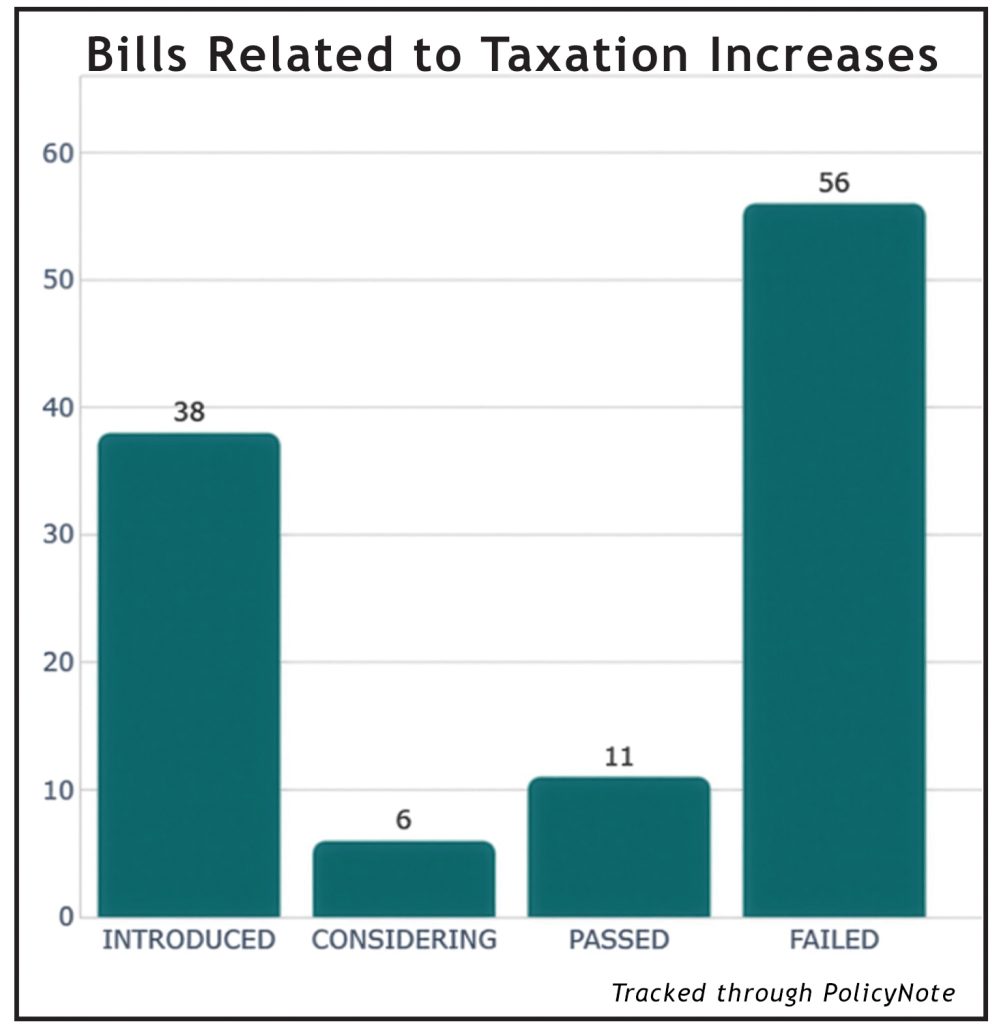
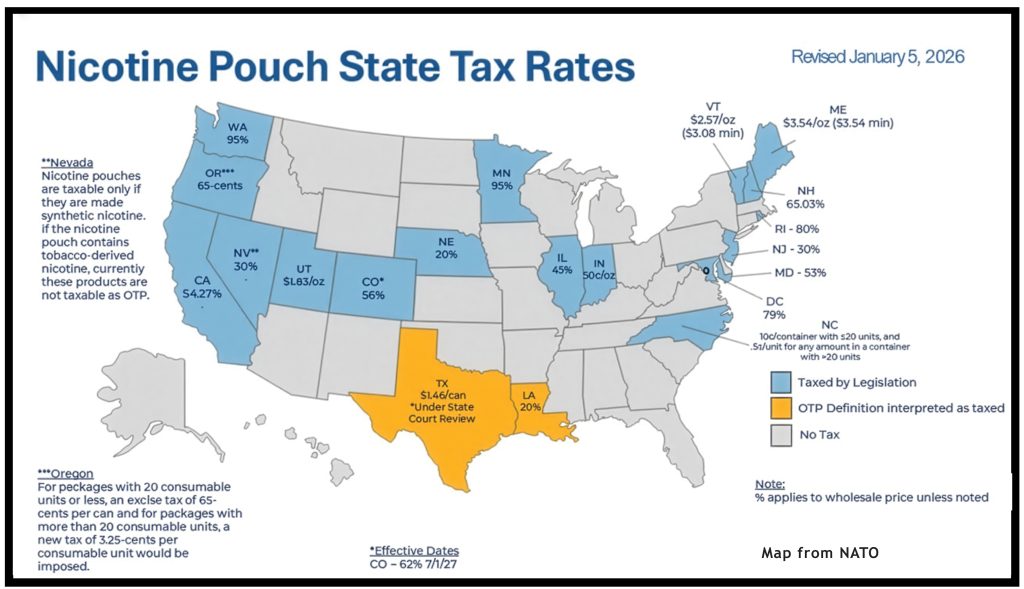
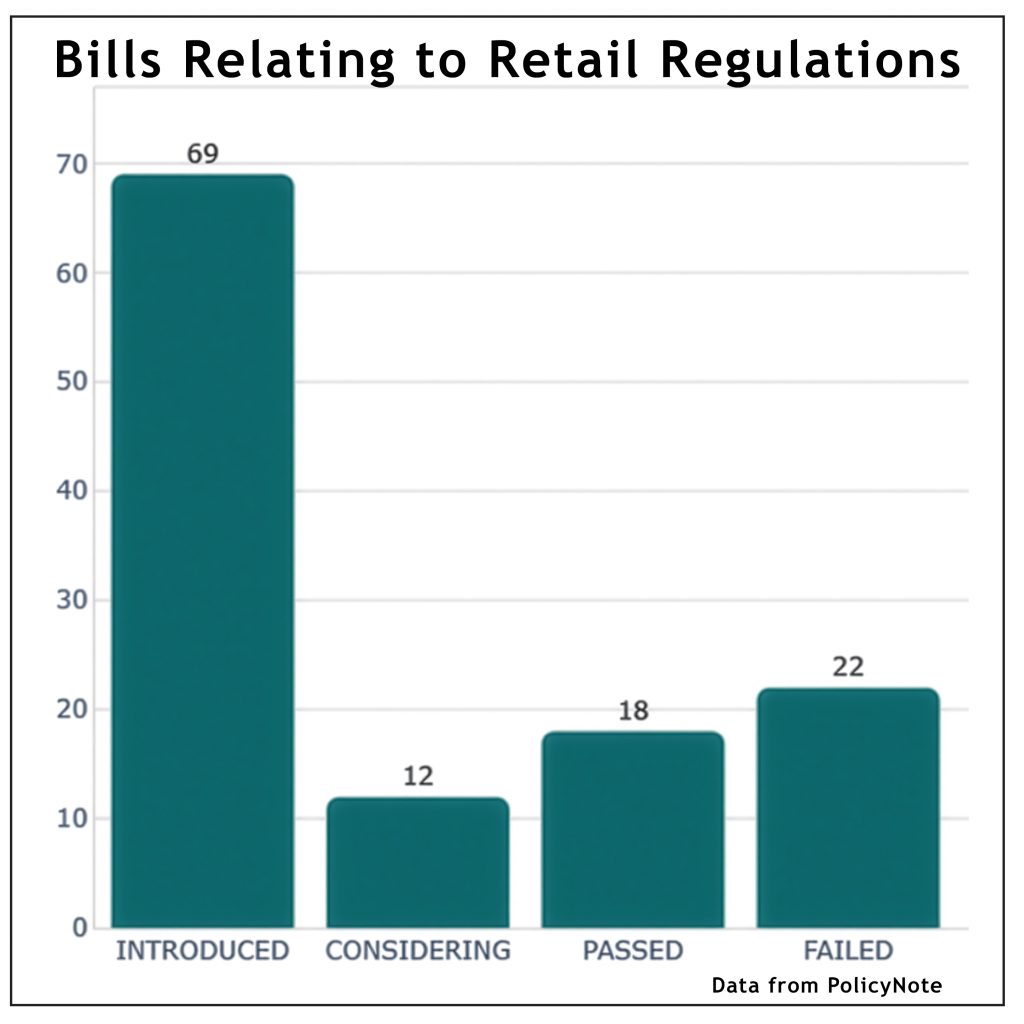
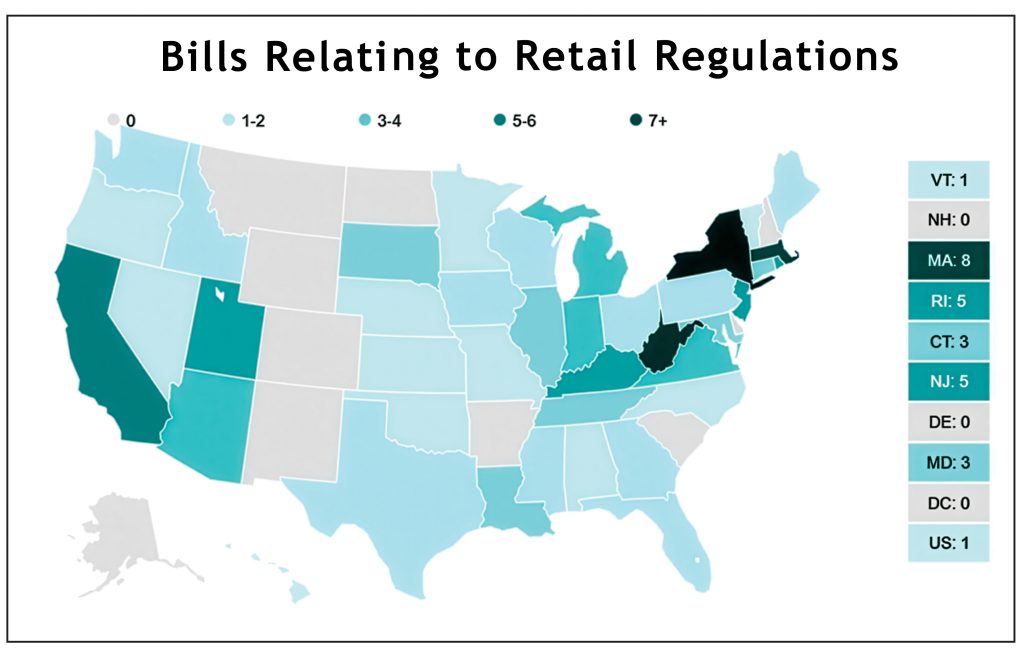
~ What FDA approvals mean for ENDS flavoured tobacco products ~
The FDA’s decision to authorise flavoured Electronic Nicotine Delivery System (ENDS) products marks one of the most significant regulatory developments the sector has seen in recent years. While tobacco and menthol authorisations had already signalled a gradual shift in regulatory thinking, the approval of mango and blueberry flavoured products suggests the agency is increasingly willing to assess reduced-risk nicotine products through a broader public health lens. Here, Chris Allen, CEO of nicotine testing and regulatory consultancy Broughton, explains the significance of this decision and why it reflects a growing emphasis on evidence-based harm reduction, behavioural science and product safeguards.
Central to the FDA’s assessment process is its “Appropriate for the Protection of Public Health” (APPH) standard, which requires regulators to weigh the potential benefits for adult smokers against the possible risks associated with youth uptake. Emerging evidence suggests that flavoured alternatives may help some adult smokers move away from combustible cigarettes, particularly those who do not successfully transition using tobacco-flavoured products alone.
One longitudinal study published via the National Library of Medicine found that adult users of sweet or fruit-flavoured nicotine vaping products were more likely to transition away from cigarette smoking than those using tobacco-flavoured alternatives.
In draft guidance published in early 2026, the FDA acknowledged that non-tobacco flavoured ENDS products may, “in certain circumstances”, support adult smokers in switching away from combustible cigarettes or increasing quit attempts. However, ENDS products continue to face heightened scrutiny due to historic concerns surrounding youth access, abuse liability and the toxicological considerations associated with inhaled products.
Importantly, the latest authorisations should not be interpreted as a relaxation of regulatory expectations. If anything, they reinforce the extent to which manufacturers are now expected to integrate public health and compliance considerations into product development at an early stage. For ENDS products in particular, this means fully characterising products from a toxicological perspective, understanding behavioural risk factors and demonstrating that appropriate controls are built into the product itself.
The FDA’s continued scrutiny of inhaled nicotine products – particularly flavoured variants – means manufacturers must increasingly show not only that products can support adult smokers in moving away from combustible cigarettes, but also that meaningful steps have been taken to restrict youth access and minimise unintended use. This will likely place greater emphasis on access-control technologies and submission-ready behavioural data as regulators continue to refine their expectations around next-generation nicotine products. For developers, regulatory strategy can no longer be treated as a final-stage compliance exercise; they must be incorporated into product design from the outset.
The latest authorisations may also have implications beyond the US market. While regulatory approaches continue to vary globally, the FDA’s decision reflects a broader shift towards more nuanced, evidence-led discussions around harm reduction and the role alternative nicotine products may play in supporting smoke-free ambitions. Increasingly, the debate is moving away from whether reduced-risk products should exist at all, and towards how they can be appropriately regulated to maximise public health benefits while minimising unintended consequences.
Ultimately, the significance of these authorisations extends far beyond the approval of two flavoured products. They signal that regulators are willing to consider the full public health picture – including adult switching potential, behavioural evidence and technological safeguards – when assessing alternative nicotine products. For manufacturers that raises the bar for scientific substantiation, product stewardship and regulatory preparedness. However, it also demonstrates that innovation supported by robust evidence and responsible product design can still find a pathway through even the most rigorous regulatory environments.
As the regulatory environment for nicotine products continues to evolve, manufacturers face growing pressure to navigate increasingly complex submission requirements across multiple international markets. Broughton supports businesses through the product lifecycle, combining scientific expertise with regulatory insight across ENDS, nicotine pouches and other alternative nicotine products. To find out how Broughton can help support your submission, visit its website.